Introduce efficiency to every step of registry management
Patient registry software built for real-world clinical workflows
The RAYLYTIC Platform is a patient registry software platform designed for hospitals, clinical research teams, and specialty registries that require seamless clinical integration, automated workflows, and secure longitudinal follow-up.
- Browser-based or clinic integration
- Audit-ready
- GDPR- and HIPAA-compliant
What Makes Our Patient Registry Software Different
Patient enrollment, form completion, and follow-ups happen automatically as part of routine care, saving time while ensuring complete, high-quality data collection.
Patient data always remains absolutely secure. Automated patient surveys, clinical data capture forms (eCRFs), and follow-ups run through your own infrastructure. Full GDPR compliance with complete institutional control.
Configure registries with drag-and-drop forms. No coding required. Real-time dashboards deliver actionable insights that drive immediate quality improvements and better patient outcomes.
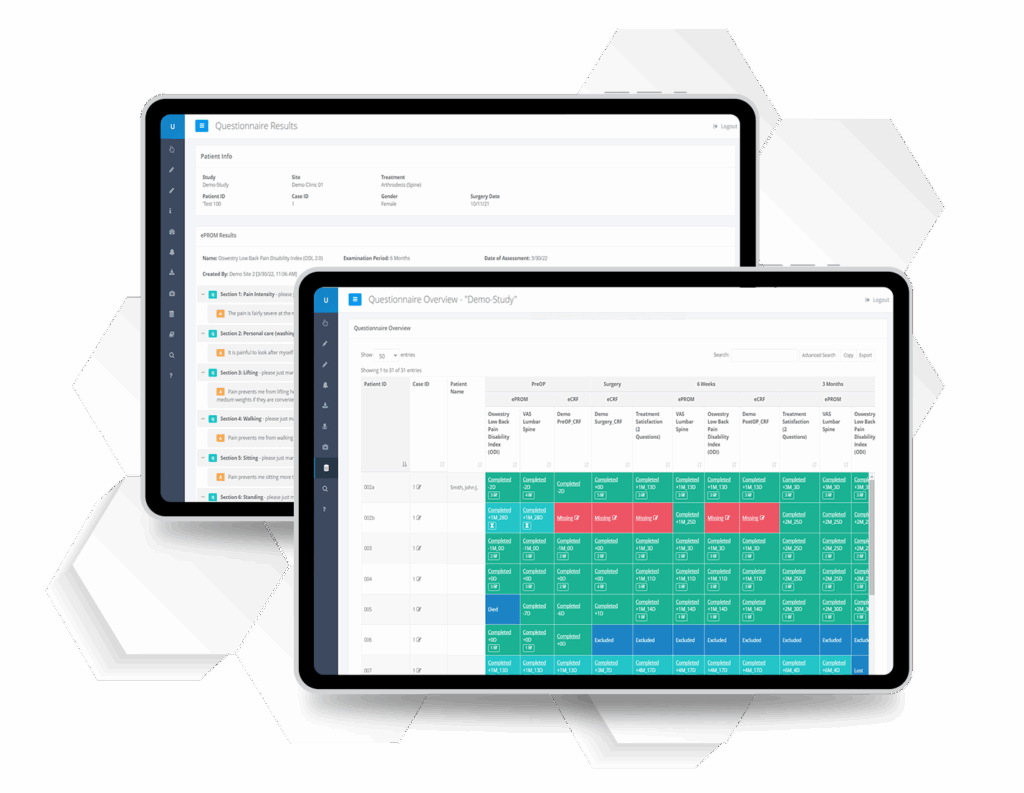
Our Patient Registry Software Seamlessly Embeds in Your Clinical Workflow.
Unlike generic survey tools or EDC platforms, the RAYLYTIC Platform embeds in routine care to capture highly contextualized clinical and outcome data along the patient journey.
- Consolidates data capture forms, secure portal delivery, and feedback analytics into your clinical routine.
- Support for descriptive statistics
- Integrated Class I Medical Device for capturing PROMs
- Export capabilities, including to national registries
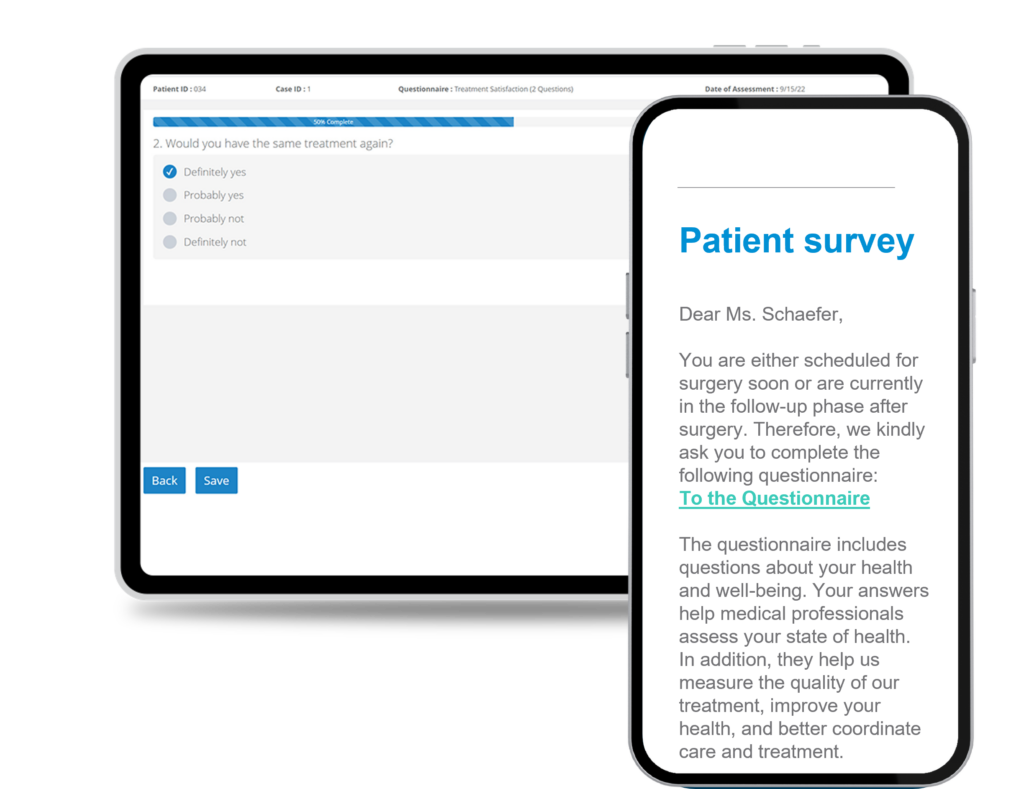
Secure, GDPR-Compliant, Hospital-Controlled Follow-Up
Long-term registries depend on consistent follow-up, but manual outreach burdens study nurses. RAYLYTIC automates patient communication while keeping identifiable data under hospital control.
- PHI remains fully within hospital access policies
- Follow-up e-mails sent from your hospital domain
- Consistent patient experience thanks to whitelabeling option
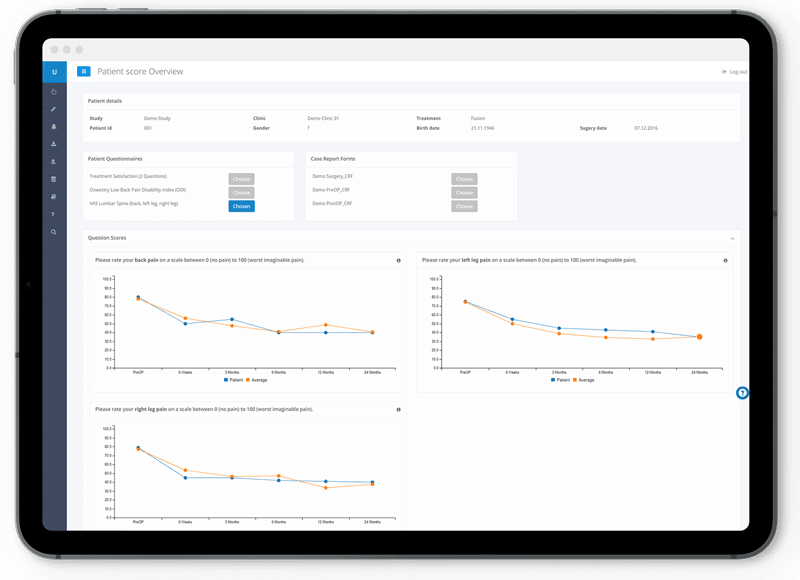
Actionable Quality Insights from Unified Data
Real-time dashboards and reports turn clinical data and outcomes into immediate tools for clinical decision-making, pathway optimization, and performance benchmarking.
- PHI remains fully within hospital access policies
- Patient-, cohort-, or clinic-level analytics (including cross-site performance analytics)
- Integrated descriptive statistics packages
Patient-reported data collection in clinical routine
Our users regularly achieve follow-up response rates of over 80%.
Turn clinical events into registry actions.
Detect relevant encounters, procedures, and diagnoses automatically. Then trigger enrollment, forms, and follow-ups aligned with your clinical protocols.
Start your hospital registry today.
RAYLYTIC turns routine care into your most valuable evidence asset. We’ll help you configure triggers, forms, and reports tailored to your workflows.
FAQs about patient registries
What is a patient registry?
A patient registry is a structured repository of clinical data that uses observational research methods to answer questions about patient health conditions, treatments, and outcomes. It collects detailed information on patients’ diagnoses, treatments, and responses over time, often focusing on specific diseases or conditions. By organizing and analyzing this data, patient registries help researchers, healthcare providers, and policymakers understand trends, improve care strategies, and facilitate medical advancements. Patient registries are crucial tools in improving patient outcomes, conducting clinical trials, and supporting evidence-based medicine.
What is the difference between EHR and a patient registry?
The main difference between an Electronic Health Record (EHR) and a patient registry lies in their purpose and use of clinical data. An EHR is primarily used by healthcare providers to document a patient’s medical history, track current clinical findings, and manage ongoing care. It focuses on individual patient care and is part of daily clinical practice. In contrast, a patient registry is designed for research purposes, collecting and managing data from a group of patients to study specific conditions, treatments, or outcomes. While an EHR supports personalized healthcare, a patient registry helps researchers analyze patterns and improve population-level health outcomes.
What is the difference between a study database and a patient registry?
The difference between a study database and a patient registry lies in their purpose, structure, and use of data.
A study database is typically created for a specific research project and contains data collected from a defined group of participants. It is designed to support a particular research question or hypothesis, and the data is often collected prospectively, as part of a controlled study or clinical trial. Study databases are usually time-limited and are specific to a single study.
On the other hand, a patient registry is a more long-term, ongoing collection of health-related data from patients, often focused on a particular disease, condition, or treatment. Patient registries are designed to track health outcomes over time and may include both retrospective and prospective data. They are typically used for observational research, public health monitoring, and improving care for specific patient populations.
In summary, while a study database focuses on a specific research question and is often part of a defined study, a patient registry is an ongoing resource that collects data to improve understanding of diseases or treatments across broader populations.