Imaging Core Lab Services for Medical Device Trials
RAYLYTIC delivers advanced radiographic analysis and fit-for-purpose imaging core lab services to aid medical device manufacturers and CROs in generating high-quality, regulatory-grade evidence.

Discover Our Imaging Core Lab Services
Designed to work seamlessly alongside the RAYLYTIC Platform, our core lab services deliver the scientific, clinical, and operational expertise your study needs for success. Trusted by over 30 medical device manufacturers across diverse regulatory pathways, we bring proven experience to every trial we support.
What We Offer
Imaging Endpoint Definition & Standardization
We support you in defining the measurement methods that lead to your imaging endpoints, and advise you on the selection of relevant parameters. In doing so, we ensure that the endpoints are clinically meaningful, measurable, and justifiable for your specific study — grounded in current literature and established standards. Please note: We do not develop study protocols or regulatory documentation, and we are not involved in direct communication with regulatory authorities.
AI-Based Quantitative Radiographic Analysis
Extract quantitative imaging endpoints, device performance metrics (fusion, subsidence, alignment), and morphoanatomical measurements with precision. Our AI-assisted tools accelerate analysis by 50% compared to traditional manual workflows while maintaining accuracy and reproducibility.
Radiologist Network for Qualitative Image Analysis
Independent blind central image review conducted by experienced, board-certified radiologists. Direct access to senior experts—not ticket queues or account managers. Our network ensures unbiased evaluation with standardized protocols and responsive, personalized support.
Image Quality & Site Compliance Monitoring
Every image is reviewed to ensure consistency in quality and protocol compliance. Real-time, proactive feedback to sites prevents costly downstream issues—unlike traditional reactive approaches that discover problems only after expensive rework is required.
Clinical Trial Imaging Infrastructure
Proprietary image management system with native EDC integration—imaging data lives within your study database, not in separate silos, including imaging eCRFs. No time-consuming reconciliation or manual data transfers. Full audit trail and regulatory compliance built in.
Imaging Review Charter Development
Agile charter development tailored to your trial's unique needs—imaging modalities, assessment methods, and protocols for ethics/regulatory submission. Based on literature review and regulatory guidance, with flexibility to adapt mid-study without enterprise bureaucracy.
We are your imaging experts for orthopedics and spine trials.
Trusted by leading MedTech companies










Radiographic Analysis for Spine and Orthopedic Trials
We provide comprehensive radiographic analysis across all major orthopedic anatomies, utilizing advanced imaging techniques to deliver precise, regulatory-grade measurements for your clinical studies.
Spine
- Range of Motion (RoM)
- Center of Rotation (CoR)
- Disc height
- Anteroposterior instability
- Cobb Angles
- Cervical & Lumbar Lordosis
- Segmental motion analysis
- Fusion assessment
- Implant subsidence
- And over 100 additional parameters
Hip
- Cup anteversion
- Cup inclination
- Pelvic obliquity
- Leg length difference
- Caput-Collum-Diaphyseal Angle
- Femoral offset
- Component positioning (6 degrees of freedom)
- Polyethylene liner wear
- Femoral stem subsidence
- Acetabular cup migration
- Osseointegration assessment
- Radiolucency detection
Knee
- Hip-Knee-Ankle (HKA) alignment angles
- Implant component positioning (6 degrees of freedom)
- Component alignment (coronal & sagittal)
- Leg alignment
- Femoral-Tibia Angle
- Joint line conversion angle
- Distal Tibia Angle
- Proximal femur angle
- Polyethylene wear quantification
- Implant migration/subsidence
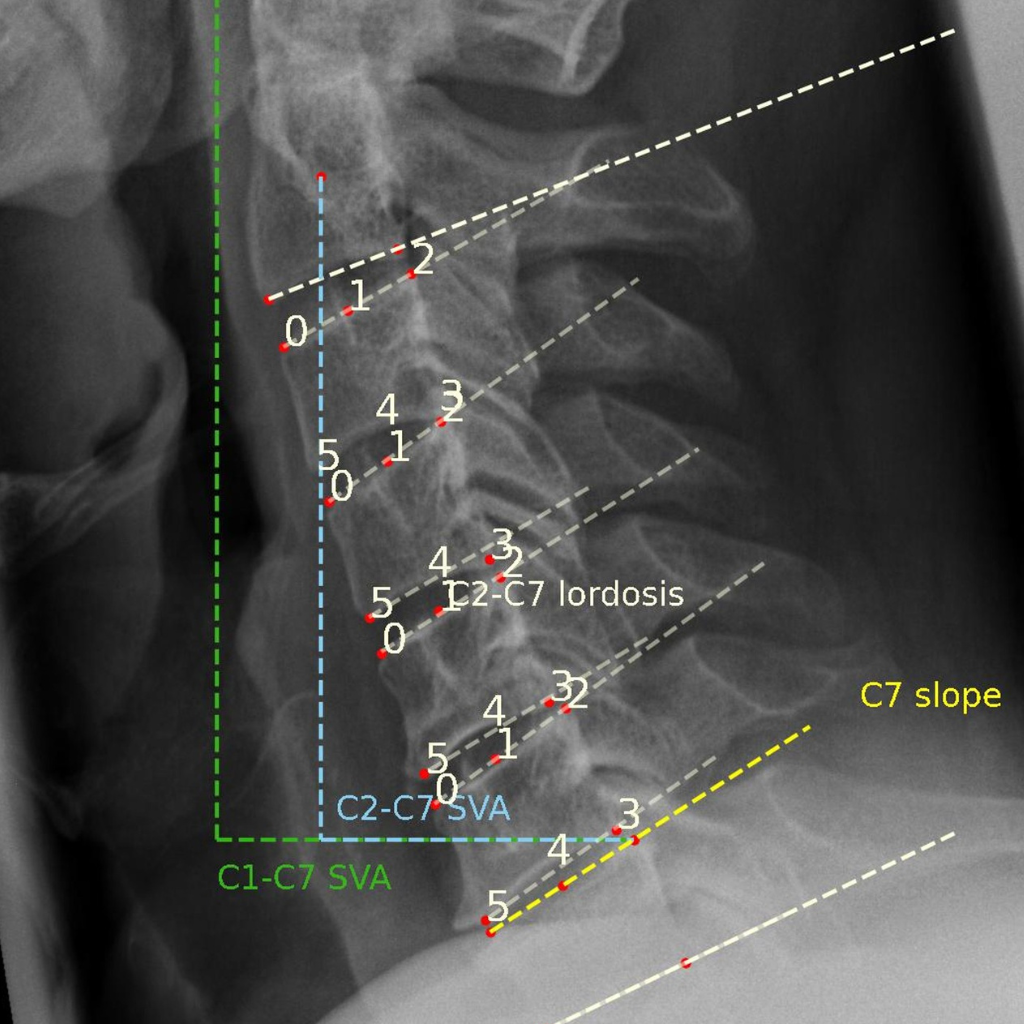
Spinal image analysis for clinical trials, registries, and research.
We have extensive experience designing and executing clinical studies with imaging endpoints for all major spinal therapies, including fusion, motion preservation, and deformity correction—from early feasibility through post-market surveillance.
Our spinal fusion experience spans cervical and lumbar procedures with allograft, autograft, and bioactive implant materials (hydroxyapatite, demineralized bone matrix), utilizing standardized protocols across X-ray, CT, and MRI modalities.
We support IDE, 510(k), and PMA submissions with regulatory-grade measurements of fusion status, segmental motion, alignment, subsidence, and device performance—all delivered with faster turnaround and seamless EDC integration compared to traditional core labs
Results presented at
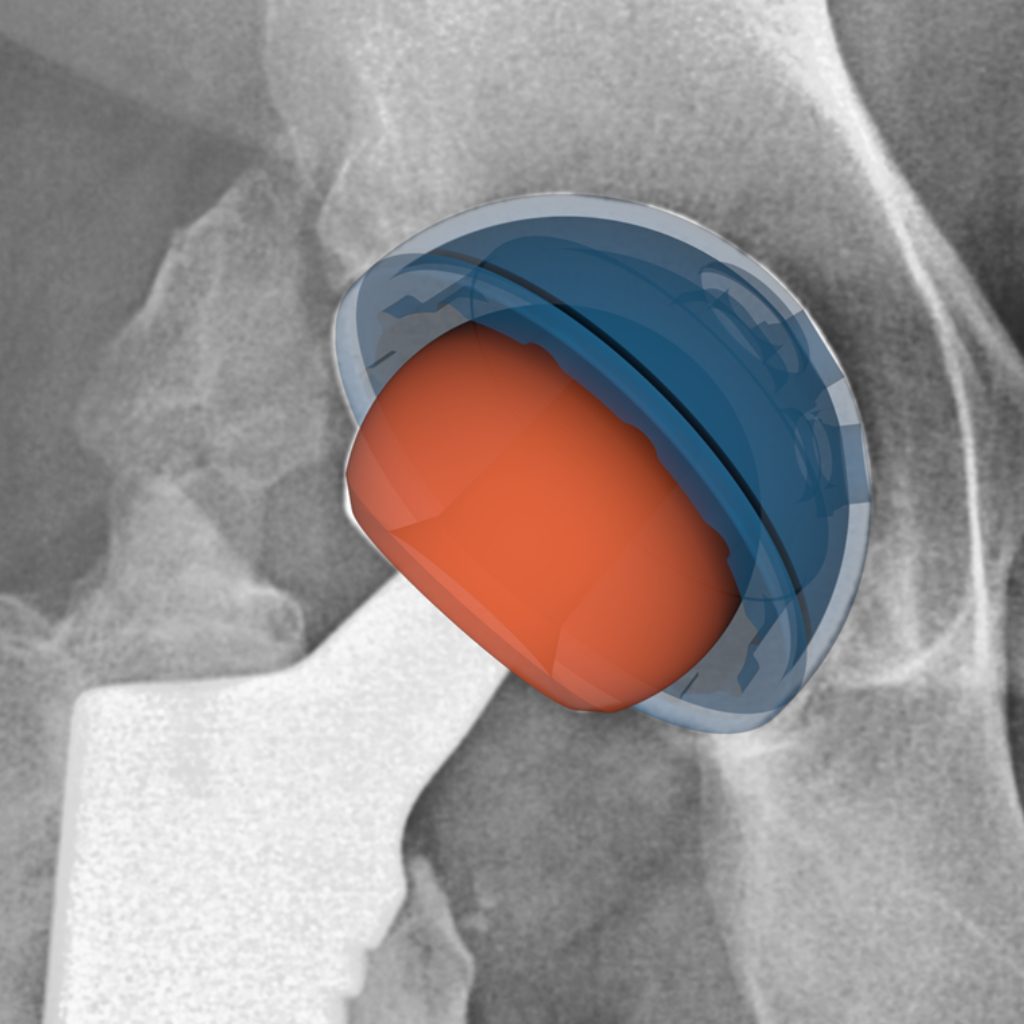
Hip implant component positioning and performance analysis.
We have extensive experience supporting total hip arthroplasty (THA) device manufacturers with radiographic analysis throughout product development—from primary THA through revision procedures and across post-market surveillance.
Our team has developed and validated proprietary 2D-3D image registration techniques that extract precise implant positioning metrics (acetabular cup inclination and anteversion) and linear wear measurements from standard clinical radiographs. This methodology has been applied across multiple clinical studies supporting regulatory submissions, enabling device safety and effectiveness assessments without specialized imaging protocols.
Our THA expertise spans:
- Primary and revision arthroplasty procedures
- Component positioning analysis for conventional and robotic-assisted surgeries
- Long-term migration assessment (femoral stem subsidence, acetabular cup migration)
- Polyethylene liner wear quantification
- Osseointegration evaluation of acetabular components
- Device integrity monitoring and radiolucency detection
We’ve supported THA device manufacturers in generating robust, regulatory-grade evidence across diverse implant designs, bearing surfaces, and surgical approaches.
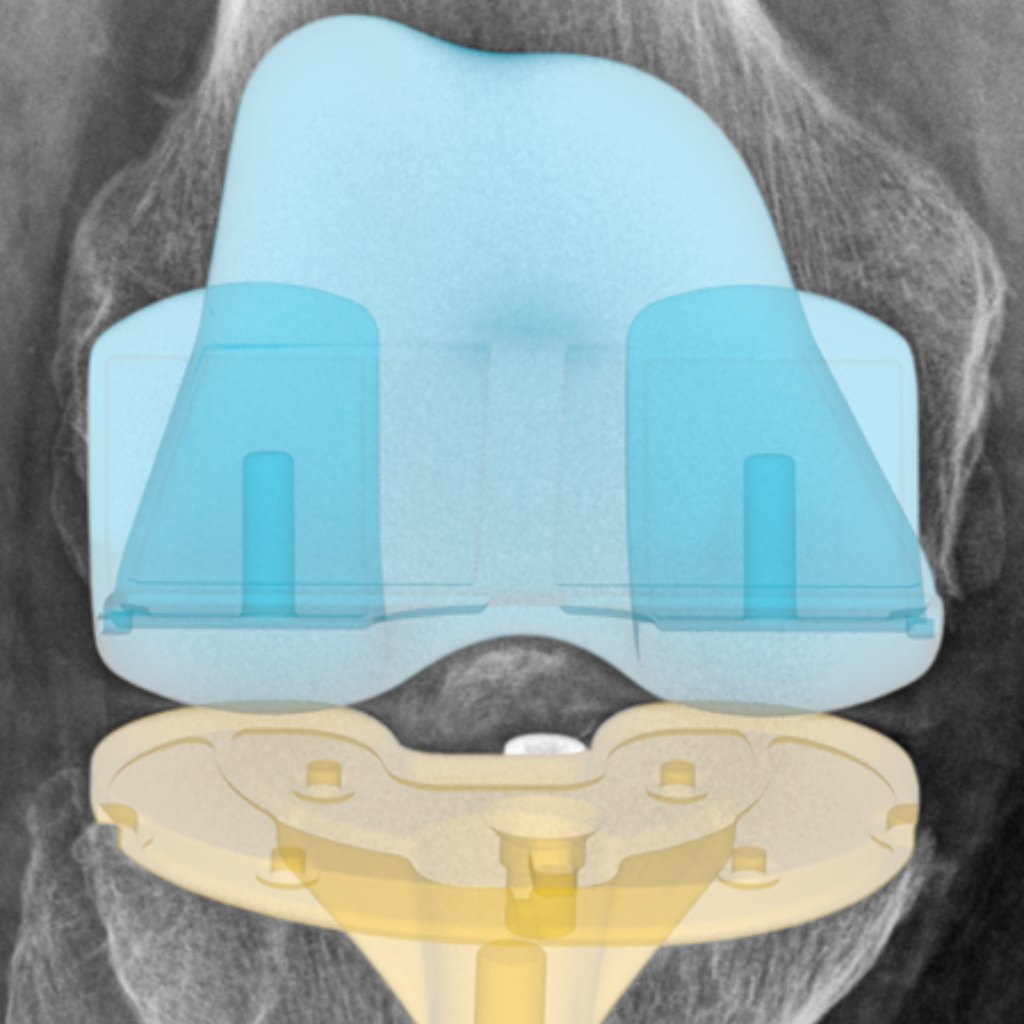
Knee implant positioning and performance analysis.
We have extensive experience supporting total knee arthroplasty (TKA) device manufacturers with comprehensive radiographic analysis throughout product development—from primary TKA through revision procedures and post-market surveillance.
Our validated methodology enables precise quantification of critical biomechanical and performance endpoints from standard clinical radiographs, including:
- Alignment assessment: Hip-knee-ankle (HKA) angle, mechanical and anatomical leg alignment
- Component positioning: 6 degrees of freedom (6-DoF) analysis for femoral and tibial components
- Component alignment: Coronal and sagittal plane positioning relative to anatomical landmarks
- Performance monitoring: Polyethylene wear quantification and implant migration/subsidence tracking over time
This approach has been applied across multiple clinical studies supporting regulatory submissions for both conventional and robotic-assisted TKA procedures, providing the regulatory-grade evidence manufacturers need across diverse regulatory pathways.
Ready to Accelerate Your Clinical Trial?
Get expert imaging analysis without the enterprise overhead. Contact our team to discuss your study requirements.
Evidence-backed expertise in musculoskeletal imaging analysis for clinical trials
Our clinical and scientific experts are leaders in the field of musculoskeletal image analysis who regularly partner with key opinion leaders in the field to validate our techniques and author peer-reviewed literature.
Spine
Can artificial intelligence support or even replace physicians in measuring sagittal balance? A validation study on preoperative and postoperative full spine images of 170 patients // read more
Hip
Cup positioning and its effect on polyethylene wear of vitamin E- and non-vitamin E-supplemented liners in total hip arthroplasty: Radiographic outcomes at 5-year follow-up // read more
Methodologies to Evaluate Linear Penetration and Volumentric Wear in Clinical Studies – Imaging Techniques for the Evaluation Of Artriculations in THA, TKA to Assess Wear and Function of the Joint Throughout the Expected Implant Lifetime // read more
Knee
What experts say about RAYLYTIC
“Special, validated, computer-assisted methods, such as Functional X-ray Analysis (FXA, RAYLYTIC, GmbH), can be used to substantially increase the accuracy and precision of evaluating implant performance."
– 1st EFORT European Consensus on Medical & Scientific Requirements for
the Introduction of Artificial Joint Arthroplasty Devices
Sample radiographic analyses
Medical image analysis for clinical trials in orthopedics and spine
The FDA-approved Functional X-Ray Analysis software (FXA) of our imaging core lab is capable of automatically evaluating over 150 radiographic parameters of the spine, hip, or knee. The reproducibility of the measurements is on file with FDA 510(K).
Explore some of our most popular automatically assessable quantitative radiographic parameters below.
Spine
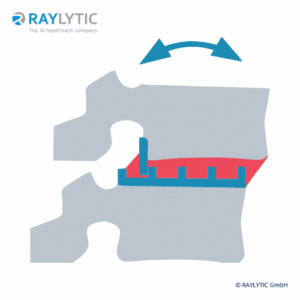
Range of Motion (RoM)
The Angular Motion/Range of Motion (RoM) is a quantitative parameter used to evaluate spinal fusion.
It is measured as the angular difference of two adjacent vertebrae during the flexion and the extension posture of a patient and is reported as an absolute value in degrees (°). RAYLYTIC’s FXA software has been shown to measure RoM 15 times more precisely than experienced physicians (Schulze et al., J Biomech 2011).
Type
Quantitative
Image type
Lateral Flex/Ex
Unit
Degrees (°)
Spine
Translational AP Motion
A further quantitative parameter used to assess spinal fusion, translational AP motion or instability is expressed as the difference in listhesis in flexion minus listhesis in extension.
Values are given in millimeters or in degrees. Positive values indicate spondylolisthesis, while negative values indicate retrolithesis.
Type
Quantitative
Image type
Lateral Flex/Ex
Unit
Degrees (°) or mm
Hip
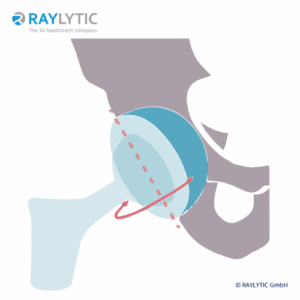
Cup Inclination and Anteversion
Cup incliniation refers to the angle in degrees (°) of the cup surface against the horizontal (medio-lateral) axis.
Cup anteversion is the angle in degrees (°) of the cup surface against the cup symmetry axis in the frontal plane.
To assess these parameters, CAD models of the implants are required.
Type
Quantitative
Image type
AP Pelvis
Unit
Degrees (°)
Hip
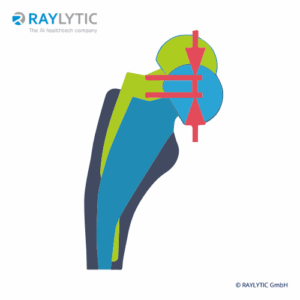
Hip Implant Migration
Migration of the hip stem relative to the femur is measured cranial-caudal (mm) and medial-lateral (mm). Angular change is measured in degrees.
Migration of the hip cup relative to the pelvis is also meausered cranial-caudal (mm) and medial-lateral (mm).
Typ
Quantitative
Bildtyp
Lateral Flex/Ex
Unit
mm
Knee
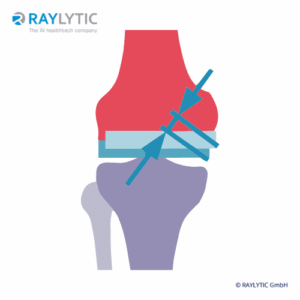
Implant wear
Wear is a key factor affecting the lifespan of orthopedic implants and refers to the gradual erosion and subsequent production of microparticulate debris, which can cause aseptic loosening.
In the past, quantifying wear has required additional invasive procedures. RAYLYTIC uses a validated, highly accurate 2D-3D registration technique to determine wear in standard clinical X-rays of knee replacements (Lutter et al. 2021, German Congress of Orthopaedics and Traumatology).
Type
Quantitative
Image type
AP Knee
Unit
mm
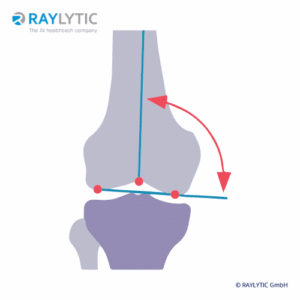
Knee
Lower limb alignment
The assessment of the knee alignment using standing full-leg radiographs (FLR) is an established and standardized method for using weight-bearing radiographs to determine the load-bearing axis of the leg (Erne et al., Diagnostics 2022).
Type
Quantitative
Image type
AP Full Leg
Unit
Degrees (°)
Planning a study?
Get expert imaging analysis without the enterprise overhead. Contact our team to discuss your study requirements.