Your images. Our expertise.
Medical Image Management Software
The RAYLYIC Platform is an AI-powered, scalable medical image management software that integrates the capture, storage, exchange, and analysis of imaging data – with unyielding compliance and security.
From compact research projects to groundbreaking clinical studies, the RAYLYTIC Platform simplifies imaging workflows by ensuring data remain compliant, consistent with study protocols, and ready for review.
Medical Image Management Built for Clinical Trials
Manage the complete medical imaging lifecycle within your clinical trial infrastructure. From automated assignment of images to visits and timepoints to central reader workflows and imaging eCRF creation, our HIPAA-compliant platform streamlines image capture, storage, and analysis.
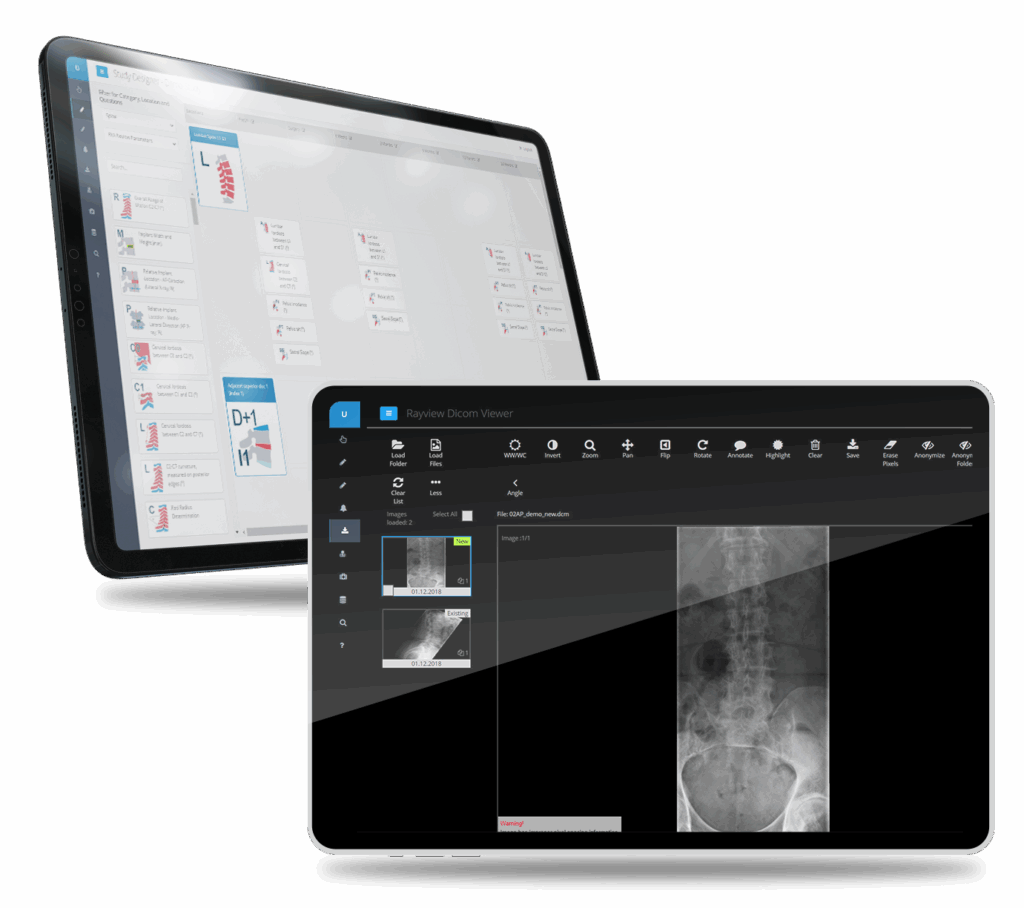
Harmonized & Secure Image Upload, Storage, and Exchange
HIPAA-compliant capture, storage, and exchange of medical imaging data with end-to-end encryption and full audit trail. Our platform harmonizes imaging workflows across sites, ensuring consistent data quality and regulatory compliance throughout your clinical trial.
Imaging eCRFs for Central Review
Create specialized imaging eCRFs that capture radiographic assessments directly in your study database. Enable central reviewers to document findings, measurements, and image quality assessments with full audit trail—eliminating the disconnect between imaging data and clinical endpoints.
Automated DICOM Timepoint Assignment
Automatically sort and assign incoming DICOM files to correct study timepoints based on protocol-defined visit windows. Intelligent matching algorithms validate patient IDs, visit dates, and imaging modalities—reducing manual reconciliation and preventing misclassified images that delay analysis.
Structured Image Capture & Storage
Secure, protocol-compliant image upload with real-time validation against imaging protocols. Images are stored with full traceability alongside clinical data, not in separate PACS or core lab portals. Sites receive feedback on image quality and protocol compliance to prevent costly downstream issues.
Central Reading Workflow
Streamlined workflow for independent central image review with blinded reader assignment, adjudication support, and real-time progress tracking. Reviewers access images directly through the platform with integrated measurement tools—no separate logins or systems required.
Native EDC Integration
Imaging data lives within your study database, not in separate silos. Image metadata, analysis results, and reviewer assessments automatically populate EDC forms with full audit trail. No manual data transfers. No reconciliation delays. Faster database lock and cleaner regulatory submissions.
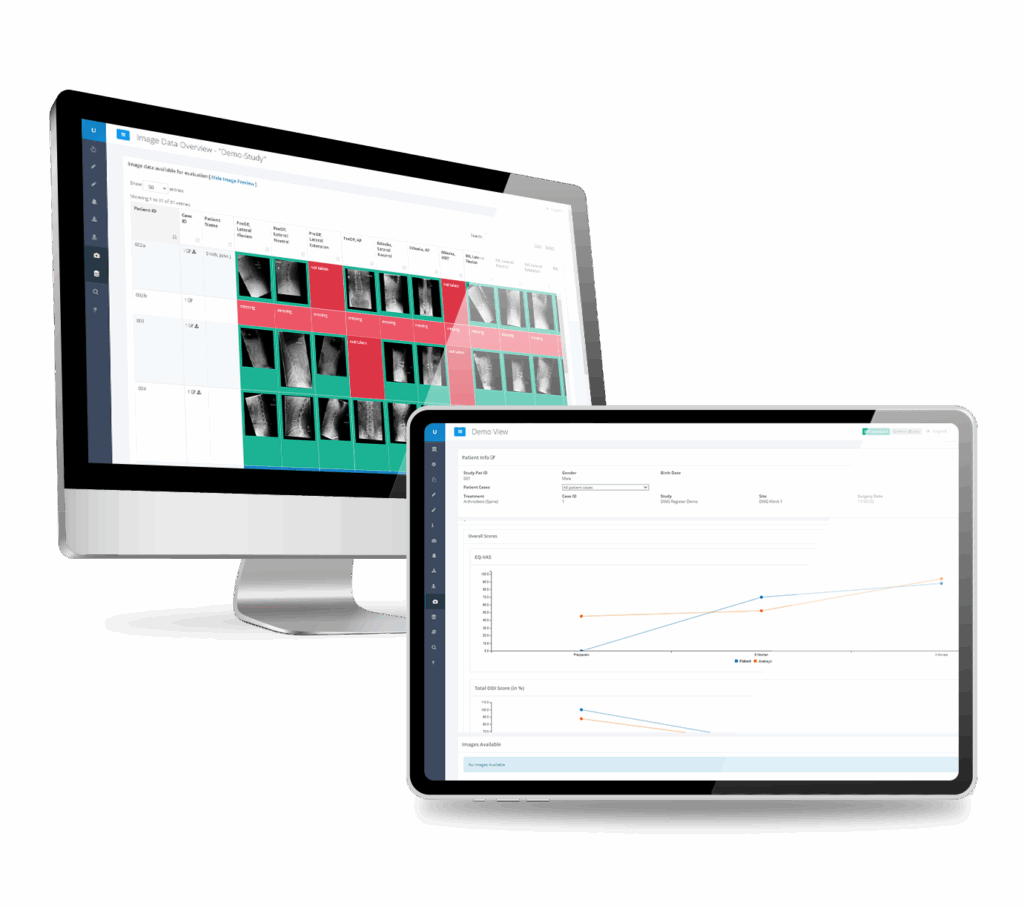
Achieve high agreement between imaging data and your study protocols.
The RAYLYTIC Platform helps sites easily upload imaging data and ensures that the images are suitable for centralized, independent image review.
Streamline your medical image management and quality control with best-in-class automation
The RAYLYTIC Platform automates QC by validating each medical image against your defined imaging specifications, minimizing errors and manual review.
Drag and drop upload of DICOM files via an integrated DICOM Image Manager
HIPAA and CFR Part 11-compliant browser-based image de-identification
Optional PACS integration
The SaaS-based platform offers a best-in-class degree of automation, including:
Image classification (e.g., anteroposterior, lateral and anatomical region)
Control of modalities and imaging types (e.g., pre- and post-operative)
Checks for duplicate images
Image suitability for analysis
Assignment of images to study examination periods
Transfer of imaging data to image reviewers based on pre-defined time points and parameters
Questionnaire designer
Customizable eCRFs, ePROs, AE forms, ePREMs, or anamnesis forms
Unscheduled AE questionnaire/form/protocol deviations
Multilingual questionnaires with no changes in user experience
Database design via drag-and-drop
Self-serve database and protocol amendments with no downtime
Unlimited database size
Ensure protocol compliance across all imaging modalities
The RAYLYTIC Platform is built to support strict study protocol compliance across every major imaging modality, including MRI, CT, PET, X-ray, and ultrasound.
Expert radiologist reviews for qualitative radiographic parameters
Integrated radiological reivew module
Blinded reviews
Adjudication workflows (resolution of discrepancies)
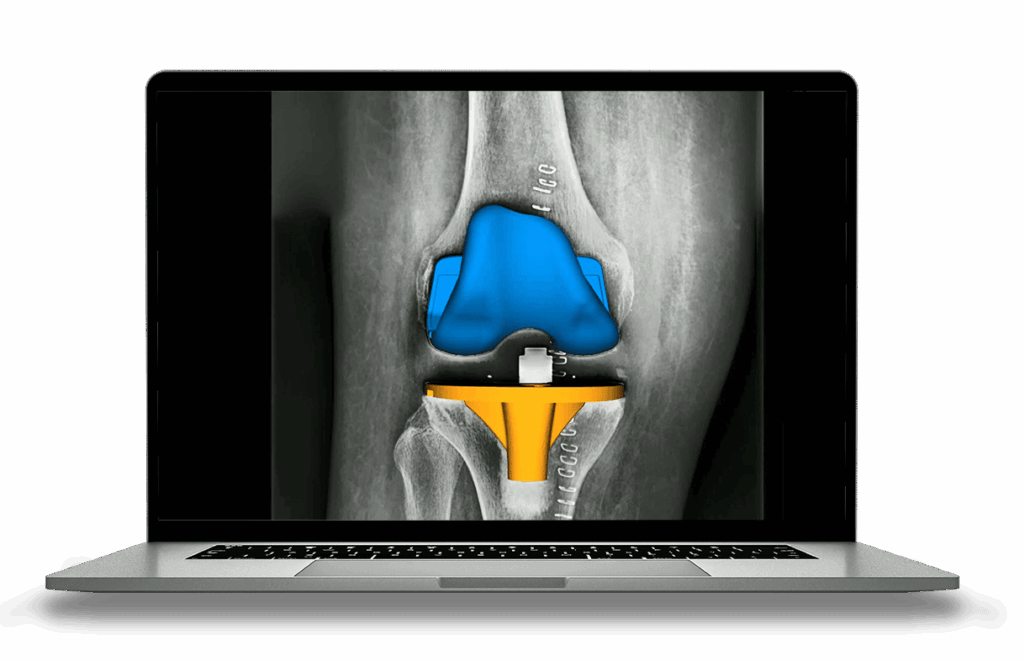
High-precision, AI-based analysis of medical imaging data for regulatory submissions
Automated assessment of over 150 radiographic parameters using FDA-certified software with consistent, sufficient, reproducible results
- Integrated AI-based image quality controls
- Real-time visibility of data completeness across sites and studies by image type or endpoint
21 CFR Part 11 compliant audit logs
Compliant with GDPR, GCP, and HIPAA
ISO 27001 and ISO 13485 certified