How Dymicron Achieved 100% Data Quality Compliance for their International PMCF Study
Streamlining complex MDR requirements across multiple sites with the RAYLYTIC Platform.
100%
Data Quality Score
40+
Hours Saved Per Site
95%
User Satisfaction
8
International Sites
Trusted by leading medical device manufacturers
The Challenge
Dymicron needed to design and execute an international PMCF study that would meet stringent MDR requirements while highlighting the unique biomechanical features of their Triadyme®-C cervical disc replacement device.
Key obstacles:
- Complex radiographic evaluation protocols
- Multi-site data coordination across borders
- Need for unprecedented biomechanical measurements
- Tight MDR compliance deadlines
The Solution
The RAYLYITC Platform combined with specialized imaging core lab services provided an end-to-end solution that transformed a complex regulatory challenge into streamlined data collection.
What made it work:
- Integrated EDC system with imaging core lab
- AI-powered image classification
- Site-friendly upload technology
- Real-time quality control
The RAYLYTIC Workflow
From setup to PMCF success in 4 steps
Design Radiographic Evaluation Protocol
Custom radiographic evaluation protocol with RAYLYTIC experts
Deploy Platform
Sites upload images automatically sorted by timepoint
Analyze Data
Imaging core lab measures biomechanical parameters
Generate Reports
Quarterly study reports
It was obvious that quality, consistency, and efficiency were the driving ideas behind each feature in the RAYLYTIC platform. The relentless focus on quality has been nothing short of impressive.
Eric Lange
VP of Research & Development, Dymicron
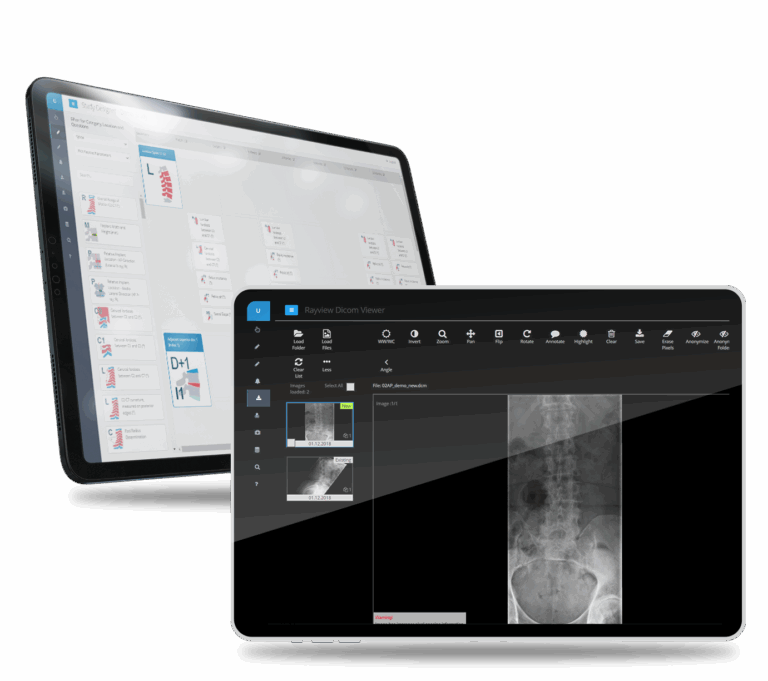
What made this possible
Precision Imaging Analysis
Never-before-measured biomechanical parameters for this type of device.
Multi-Site Coordination
Seamless data collection across international sites with no third-party software needed.
Expert Support Team
Board-certified radiologists and imaging experts conduct all measurements in-house.
Automated Image Sorting
AI-powered classification automatically organizes images to correct follow-up timepoints, saving medical staff countless hours.
GDPR-Compliant Security
Enterprise-grade data protection with full GDPR compliance built into every feature.
Real-Time Quality Control
Only complete datasets can be saved, ensuring 100% data quality from the start.
The main advantage of the RAYLYTIC Platform is the ability to automatically sort images to the correct follow-up time points. This feature saves medical staff countless hours of sorting images to different folders before upload.
Dr. Ardeshir Ardeshiri
Neuro-Spine Surgeon, Wilhelmsburger Hospital Gross-Sand, Hamburg
RAYLYTIC Platform vs. Traditional EDC Systems
| Feature | Traditional EDC | RAYLYTIC Platform |
|---|---|---|
| Integrated Imaging Core Lab | ✗ Separate vendor needed | ✓ Built-in with experts |
| AI Image Classification | ✗ Manual sorting required | ✓ Automatic timepoint sorting |
| Advanced Biomechanical Analysis | ✗ Limited parameters | ✓ Custom REP capabilities |
| Data Quality Enforcement | ✗ Post-collection cleanup | ✓ Real-time validation |
| Third-Party Software Required | ✓ Often necessary | ✗ All-in-one solution |
The Results: More than Just Compliance
High-Quality Data
Complete, validated datasets from every site with minimal post-collection cleanup needed.
Happy Users
Both clinical sites and internal teams praised the intuitive interface and seamless workflow.
Unprecedented Insights
Biomechanical measurements never before captured in this type of study.
Regulatory Confidence
Complete MDR compliance with robust safety and efficacy documentation.
Ready to Streamline Your PMCF Study?
Join Dymicron and other leading medical device manufacturers using RAYLYTIC to achieve regulatory excellence with less effort.