All your endpoints. One solution.
Electronic data capture (EDC) system
Capture clinical, imaging, and patient‑reported data in one validated platform instead of stitching together separate EDC and imaging tools. The RAYLYTIC Platform contains 21 CFR Part 11-compliant EDC functionality, specifically designed to streamline data management and ensure regulatory compliance for medical device trials.
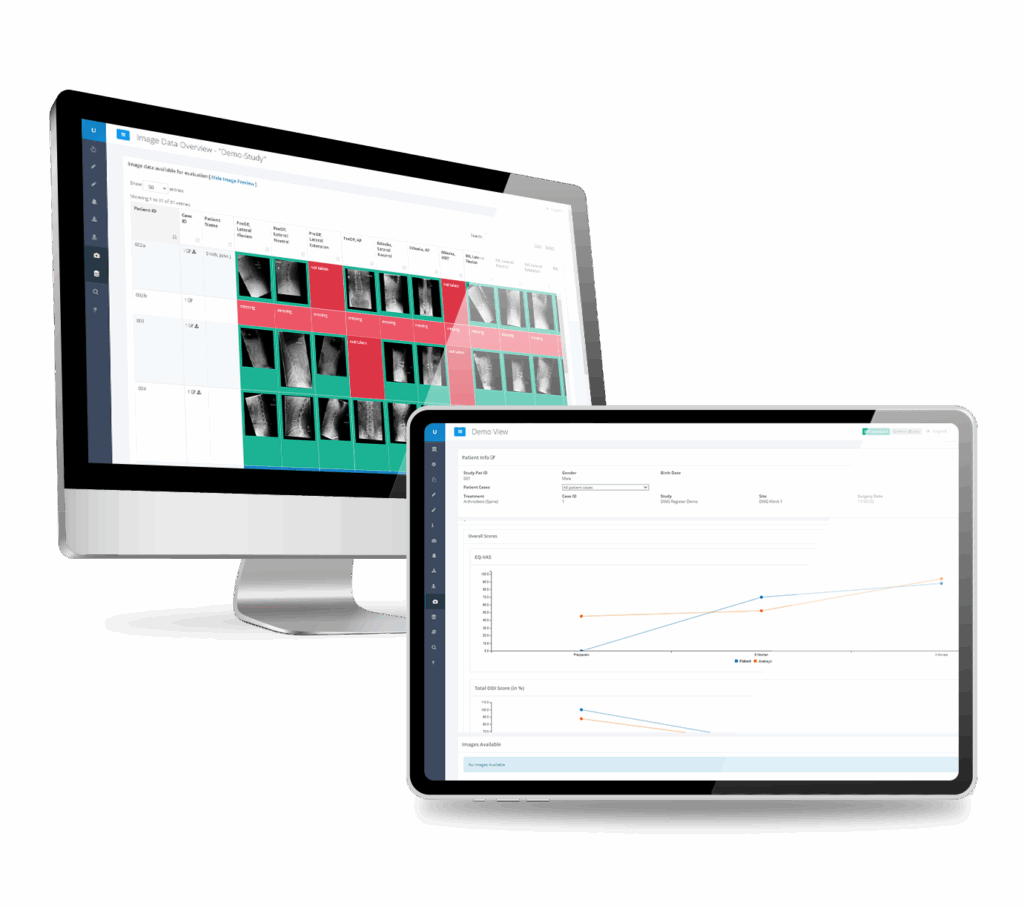
The Only EDC-System with Native Imaging Integration
Imaging endpoints shouldn’t live in separate silos. Our EDC platform natively integrates imaging eCRFs with clinical data collection, eliminating the manual reconciliation that delays database lock. Capture radiographic assessments, device performance metrics, and patient outcomes in a single system.
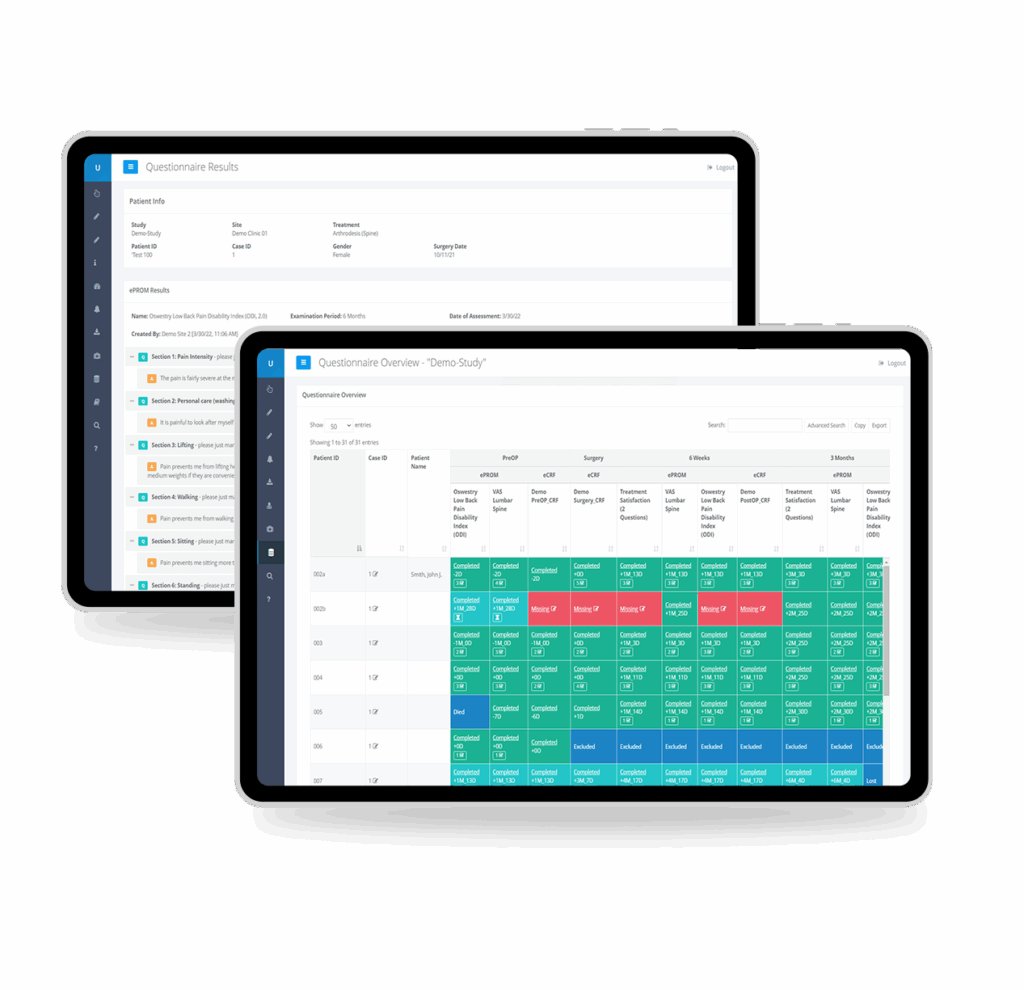
eCRFs
Design flexible eCRFs tailored for medical device trials—from standard visit forms to device-specific assessments. Built-in edit checks, automatic validation, and intuitive form design ensure high-quality data capture without programming expertise.
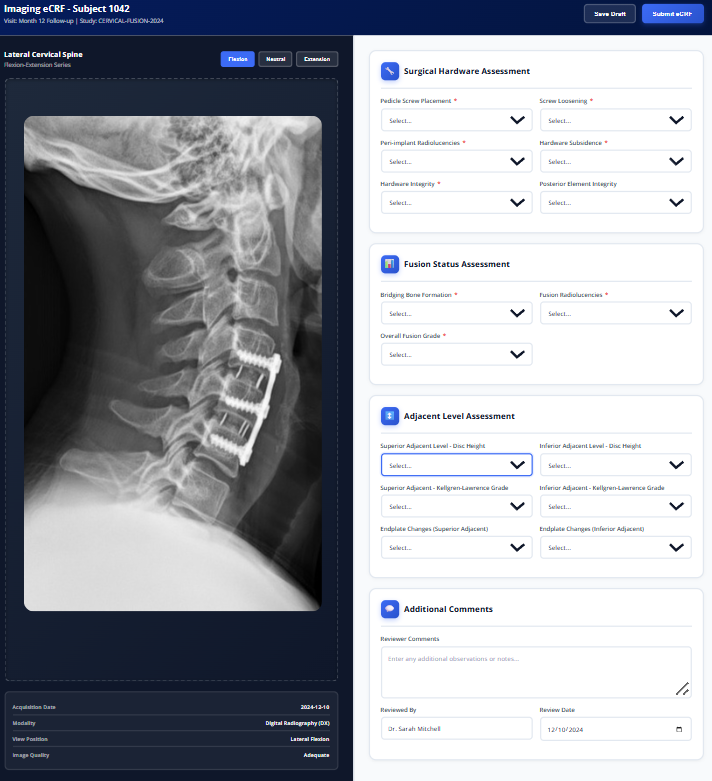
Imaging eCRFs
Purpose-built imaging eCRFs capture radiographic assessments directly within your study database. Native integration with imaging endpoints eliminates manual reconciliation, accelerates database lock, and ensures imaging data lives alongside clinical data—not in separate silos.
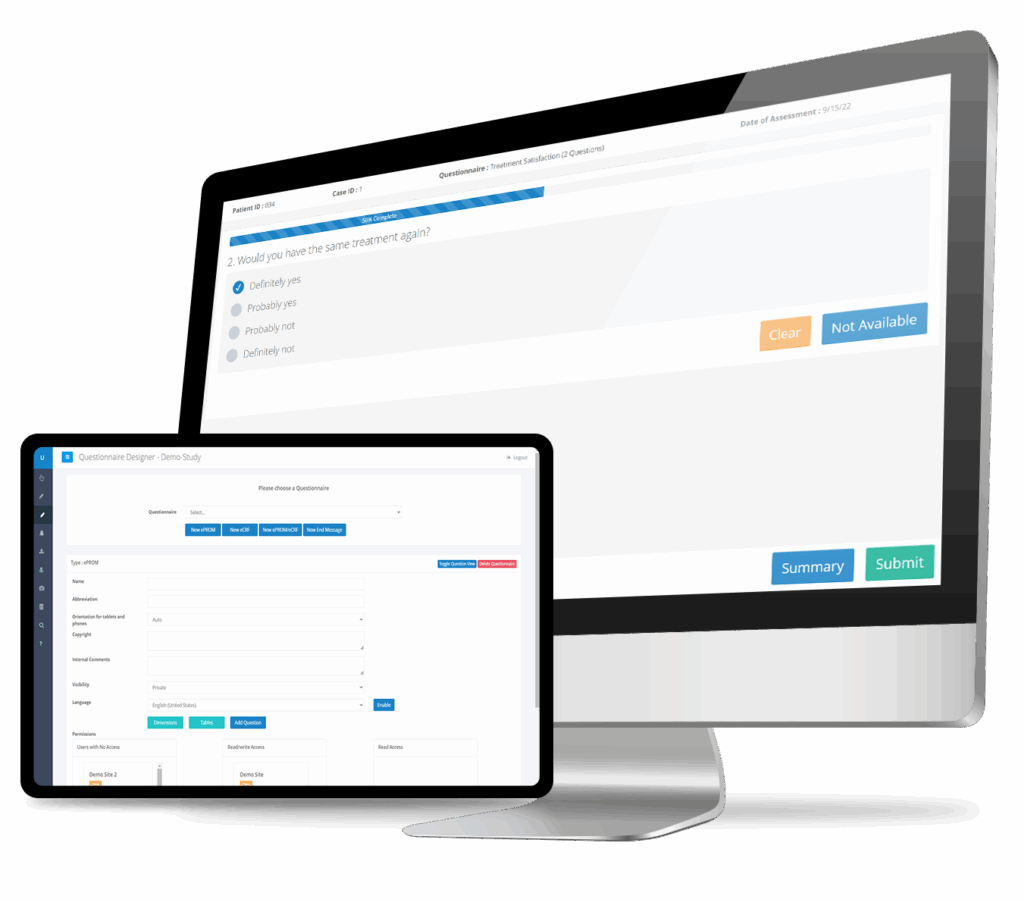
ePROs & PROMs
Full electronic patient-reported outcome (ePRO) and patient-reported outcome measure (PROM) functionality with automated scheduling, remote data capture, real-time scoring, and instant visualization. Purpose-built for device trials where patient experience and functional outcomes are critical endpoints.
Custom Questionnaires: AE Forms & More
Build custom forms for adverse event reporting, device deficiency tracking, concomitant medication logs, and any study-specific data collection needs. Our intuitive form designer lets you create regulatory-compliant questionnaires without programming—tailored precisely to your device trial requirements.
Capture clinical data and imaging data in one unified, compliant EDC system.
Bring eCRFs, imaging files, and patient outcomes together in a single source of truth to reduce manual work, data silos, and query cycles. Your teams spend less time managing systems and more time on what matters: advancing patient outcomes and accelerating study completion.
Questionnaire designer
Customizable eCRFs, ePROs, AE forms, ePREMs, or anamnesis forms
Sponsor-specific questions for logging protocol deviations in eCRFs
Unscheduled AE questionnaire/form/protocol deviations
Multilingual questionnaires with no changes in user experience
Database design via drag-and-drop
Self-serve database and protocol amendments with no downtime
Unlimited database size
- Scalable infrastructure handling images from single-site to global multi-center trials
- HIPAA-compliant image upload, transfer, storage (automatic deidentification of imaging data)
- Integrated eCRF creation for central image review
- Structured image review templates configured for your specific study protocols
- Smart image assessment to support compliance with study protocols
- Centralized imaging database with multi-site access and role-based permissions
- Support for multiple image modalities (CT, MRI, X-Ray, ultrasound, and more)
- Integrated AI-based image analysis for select radiographic outcomes
- Customizable dashboards for principal investigators, site coordinators, and trial sponsors
Generate powerful real-world evidence in a flexible EDC-system
Our EDC-system is designed to automatically capture data directly from the sources that matter most to your study.
Sponsor-initiated registries for continuous RWD collection
Internationally accepted
Compliant with EU and US data privacy regulations
Integration with popular EHR, appointment, ERP, lab, archiving softwares
Configurable integration layer to merge data between HIS systems and UNITY
Bridge between HL7 v2/3/CDA to FHIR
Bi-directional interface
Exports in neutral CSV format for easy consumption by SAS, Excel, or other applications
Profiles, mappings, and APIs to fit your master data management needs
Questionnaire designer
Customizable eCRFs, ePROs, AE forms, ePREMs, or anamnesis forms
Sponsor-specific questions for logging protocol deviations in eCRFs
Unscheduled AE questionnaire/form/protocol deviations
Multilingual questionnaires with no changes in user experience
Database design via drag-and-drop
Self-serve database and protocol amendments with no downtime
Unlimited database size
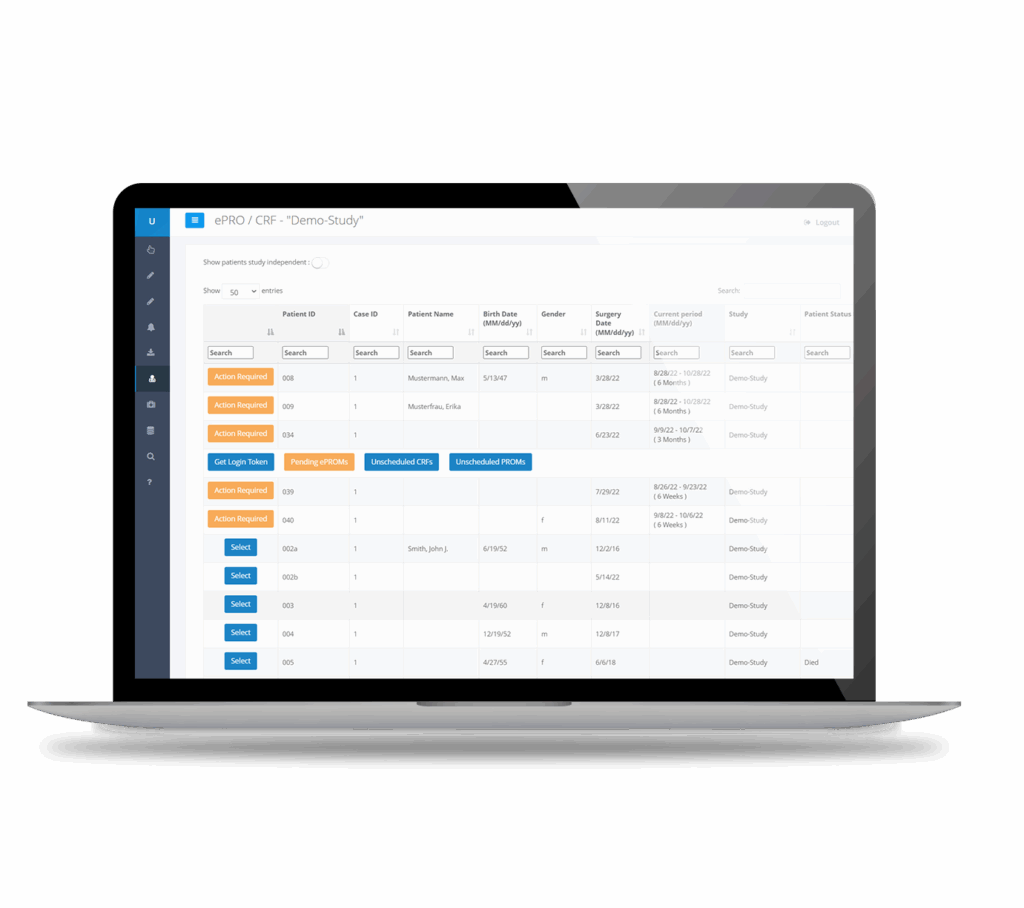
Manage multicenter studies with ease
Monitor large-scale studies with real-time reporting, adjustable alerts, and rich visualizations of study progress: Our EDC-System integrates seamlessly with our CTMS.
Automated export of ePRO data to third-party registries (e.g., SIRIS, DWG)
Real-time data quality analysis and validation
Event- or time triggered alerts, notifications, reminders, data submissions and reports to automate administrative tasks
Automated edit checks involving multiple data fields and datasets
Automated cross-check of information within and across eCRFs, ePROs, and other questionnaires
Separate environment for conducting validation
Configurable email alerts based on customizable criteria e.g.,
Advance information to sites about upcoming patient visits
Data values for certain form submissions
Study completeness notifications
Alerts based on patient reponses to questionnaire items
Initial invitation and automated reminder alerts to automate communication with patients
Dashboards showing study progress, upcoming tasks, data status
Customizeable reports on overall enrollment (per study and/or site), data completeness, notifications
Scheduled or ad-hoc reports and exports
Source data verification with variable granularity (e.g., field, page, form, patient level)
Electronic investigator and patient signatures in compliance with 21 CFR Part 11
Medical coding using standard dictionaries e.g., MedDRA, RadLex (others upon request)
Assign medical codes to verbatim terms on existent data
Auto-coding, synonym lists, detection of coding conflicts
Display of all open and resolved queries
Simple query communication and resolution process
Adjustable user interface for patients
Design and user interface automatically adjust to device
Data entry via tablet, mobile phone, upload of source and archival documents
- Comprehensive electronic data capture training
An EDC-system sites will thank you for.
The RAYLYTIC Platform automates communication between sites, sponsors, and patients to streamline your clinical study operations.
Remain compliant with confidence
The RAYLYTIC Platform is innately compliant and fulfills the most stringent regulatory demands.
The ePRO module is a class I medical product in Europe according to the European Medical Device Regulation 2017/745/EU, and the EDC module is FDA 21 CRF Part 11 compliant in the United States.
All system interactions are 100% audit-logged
Exportable audit trails in PDF format
FDA 21 CFR Part 11 compliant
GDPR-, GCP- and HIPAA-compliant
ISO 27001 and ISO 13485 certified
SaaS architecture with ISO 27001-certified data storage and ISO 13485-certified business processes




Must-knows about EDC-systems and eCOA
The use of electronic data capture (EDC) and electronic clinical outcome assessment (eCOA) for clinical trials has grown significantly in the last two decades. But as more solutions have come to market, the lines between EDC systems, eCOA, and ePRO have blurred increasingly.
What is the difference between an EDC-system and eCOA?
While often used synonymously, electronic data capture (EDC) and electronic clinical outcome assessments (eCOA) actually represent two different pathways for collecting clinical study data and integrating them into a database.
EDC data
EDC data is entered into the study database by study coordinators, clinical research associates, or data managers. These data may be collected either purely electronically or be transcribed from a paper-based case report form (CRF) into a corresponding electronic form. While these data are ultimately sourced from patients or study participants, clinical study staff are responsible for getting it into the database.
eCOA data
eCOA data, however, is collected directly from patients, doctors, or caregivers. Unlike EDC, study coordinators do not act as intermediaries for transferring the data into the database.
Another difference between eCOA and EDC is their functional complexity. Due to a CRA or data manager’s need to fine-tune the consistency and quality of data, EDC systems offer more options for querying, comparing, and reviewing data. eCOA modules, on the other hand, generally possess a more streamlined interface designed to help participants navigate the software and fill out questionnaires accurately and entirely.
The function of edit checking is one way to illustrate this difference. Edit checking in an EDC module gives a data manager more options for constructing input limitations, verifying data, and cross-checking data between different forms and questionnaires. Edit checking functionalities in an ePRO, however, are geared toward guiding the patient to fill out a questionnaire completely and accurately.